Read the Praxis Annual Report 2022
We are pleased to share the 2022 Praxis Annual Report with you, which focuses on collaboration and how it drives impact. The report summarizes the Institute’s impact on spinal cord injury research, innovation, and care, with stories showing how collaboration builds community, activates advocacy, and directs priorities.
Collaboration is critical to helping Praxis work towards its vision of a world without paralysis after spinal cord injury. It brings more hands, eyes, and brains together to drive research collectively forward for more rapid success. All of our work is built around leadership and engagement with people with lived experience of spinal cord injury.
The report is now available in both English and French.
Our Approach
Moving Knowledge into Action.
Embedding Lived Experience Expertise
At Praxis, we believe that people with lived experience of spinal cord injury play a vital role in driving and supporting the adoption of the best research and clinical practices.
Person-centered Model
We focus on the unique needs of individuals living with spinal cord injury at the heart of everything we do. From the moment of injury and through acute care, rehabilitation, and community reintegration, we work to address key challenges and improve outcomes.
Collaborative Impact
Praxis brings together leaders in spinal cord injury research, care, and innovation by connecting, convening, and collaborating to harness the power of networks to make an impact.
Transforming SCI Outcomes
Using Data
Praxis uses data as a key driver of change, developing data-driven algorithms and clinical tools. We create solutions that enhance outcomes for individuals with spinal cord injury and accelerate research.
Key Initiatives
DONATE
Make a Difference
How would you like to change the world? Imagine improving the lives of millions of people who live with a spinal cord injury.
Donate to the Praxis Spinal Cord Institute today. Your donation will forge innovations that reduce paralysis and related complications and enable individuals with spinal cord injuries to live their best lives.
Praxis News
VIEW ALL

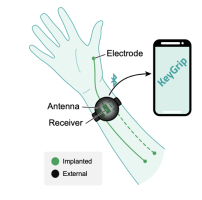
How direct engagement with users shaped KeyGrip’s path forward
For Megan Moynahan, a biomedical engineer and founder of KeyGrip, the goal was clear—develop an implantable device to restore hand function for people with cervical spinal cord injuries (SCI). The science was strong, and the need was well-documented. But one crucial question remained: How would potential users and surgeons actually perceive it?
Through the Praxis 2024 SCI Incubate Program, KeyGrip gained direct access to Persons with Lived Experience (PLEX) of SCI and regulatory experts. These conversations reshaped their messaging, reintroduced a product feature they had ruled out, and provided a clear roadmap for entering the Canadian market.
Hearing directly from users changed everything
KeyGrip’s initial assumption was that anyone with SCI seeking more independence would want to improve their hand function. But through focus groups with PLEX members, they uncovered a more complex reality—potential users had concerns about surgical risks, recovery time, and the fear of losing existing function.
Rather than just highlighting the benefits, KeyGrip needed to address fears head-on, ensuring their messaging reassured users and provided clear data on the long-term impact.
“We always knew people valued hand function—but hearing their concerns in their own words made us rethink how we communicate our solution.”
Bringing a feature back into the design
One of the most unexpected takeaways was how users viewed different control methods. Initially, KeyGrip had dismissed voice activation, assuming it wasn’t a priority. But user feedback challenged that assumption.
This insight put voice control back on the table, influencing upcoming development proposals.
“People may not want to talk to their hands in public, but I was really surprised that they are open to voice activation at home, making it a viable control method once again.”
A Clearer Regulatory Pathway for Canada
Beyond product and messaging refinements, Praxis helped KeyGrip navigate the Canadian regulatory landscape, an area they hadn’t initially prioritized. Through mentorship and expert connections, they developed a clear plan for clinical trials and approvals.
This new perspective expanded KeyGrip’s market strategy, ensuring they could plan for approvals in multiple regions early on.
Insights that will shape KeyGrip’s next steps
Unlike traditional incubators with a set curriculum, Praxis adapted to what FMRK needed—whether that was regulatory guidance, market validation, or direct feedback from Persons with SCI Lived Experience (PLEX).
The biggest shift for KeyGrip wasn’t just in strategy—it was in how they approached decision-making. Conversations with PLEX members provided real-world perspectives that no market research could replicate, while regulatory guidance gave Megan and her team the clarity they needed to move forward with confidence.
“Every time I have a stakeholder conversation; it improves my thinking in a positive way. It helps me make decisions faster, with more confidence.”
One of the most profound realizations was that building an innovative product isn’t just about function—it’s about trust. People with SCI valued their independence and were naturally cautious about interventions. The challenge wasn’t just demonstrating that KeyGrip could improve hand function—it was proving that it wouldn’t compromise
what users had already adapted to.
Rather than simply accelerating their development, the greatest impact of Praxis was helping KeyGrip make the right decisions at the right time. Whether through PLEX insights, regulatory guidance, or 1:1 expert mentorships, every step brought them closer to a product that truly meets the needs of both users and clinicians.
“We need to collect the right evidence, not just to prove our technology works, but to help people feel confident in making the decision.”
About KeyGrip
KeyGrip is a simple implanted stimulation system that provides lateral pinch to people with cervical spinal cord injury, enabling them to pick up and use small objects and giving them independence in eating, writing, and other activities of daily life. KeyGrip has 2-3 implantable electrodes that can be placed in an outpatient procedure to activate paralyzed muscles. Activation of the implant is achieved through externally placed components.

 How FMRK Uncovered a Critical Gap in UTI Testing and Pivoted Fast
How FMRK Uncovered a Critical Gap in UTI Testing and Pivoted Fast
When Faisal Khan founded FMRK, he set out to reduce unnecessary antibiotic prescriptions for UTIs. However, a conversation with a Praxis alum introduced him to a critical unmet need—people with spinal cord injuries (SCI) face unique challenges with UTIs, yet existing solutions weren’t designed for them. This realization led FMRK to join the Praxis 2024 SCI Incubate Program, where they refined their product, validated their market, and made a key regulatory breakthrough that shaped their path forward.
Uncovering Real User Needs
“I knew there were issues with collecting urine samples, but I didn’t realize just how pronounced it was within the SCI community.”
Before Praxis, FMRK understood UTIs were a significant issue, but they hadn’t fully grasped the everyday challenges SCI individuals faced in managing them. Through the program’s focus groups, they engaged directly with People with SCI Lived Experience (PLEX)—a community
of individuals sharing firsthand knowledge of living with SCI. These conversations revealed major pain points, such as the difficulty of urine sample collection, an issue FMRK had initially underestimated.
This insight reshaped their product strategy, leading them to explore ways to simplify the collection process and ensure their technology was truly accessible.
Regulatory Breakthrough That Changed Everything
“That had a huge impact, it was monumental. Can’t really stress enough how big a shift that was to have a solid case… It changed our entire regulatory pathway—cost, time, everything.”
FMRK had assumed their device would be classified as a Class III medical device, requiring extensive time and resources to bring to market. But Praxis mentors helped them build a strong case for Class II classification, significantly reducing regulatory hurdles and accelerating their timeline. This shift reshaped their business strategy, impacting everything from fundraising to market entry.
Moving from Theory to Execution
Initially, Faisal planned to raise capital before advancing product design, but Praxis changed that approach.
“Instead of waiting to raise money, we decided to go straight into product development.”
Through validation with PLEX members and guidance from mentors, FMRK gained the confidence to move forward faster. By the end of the program, they had secured a partnership with UBC to begin product development—saving valuable time and pushing them closer to market launch.
Learnings from the Praxis Experience
Unlike traditional incubators with a set curriculum, Praxis adapted to what FMRK needed, whether that was regulatory guidance, market validation, or direct feedback from People with SCI Lived Experience (PLEX).
“Most programs just bring in industry leaders each week,” Faisal shared. “Praxis actually asked, ‘What do you need help with?’
and worked with us to solve those challenges.”
Engaging directly with PLEX members gave FMRK insights they wouldn’t have gained elsewhere. Instead of designing in isolation, they were able to test ideas, validate assumptions, and refine their product based on real user needs. This approach didn’t just improve their technology—it ensured their solution was practical, accessible, and valuable to the people it aimed to serve.
By the end of the program, FMRK had a clear strategy, an optimized product roadmap, and a faster path to market. More than anything, their experience reinforced a key lesson: success isn’t just about technology—it’s about understanding and designing for the people who will use it.
About FMRK
FMRK Diagnostic Technologies helps enable and advance decentralized healthcare through easy-to-use, near-patient diagnostic tests. Their flagship product delivers Personalized Early Intervention Recommendations (P.E.I.Rs) to patients with suspected Urinary Tract
Infections (UTIs) in community settings like Pharmacies and Long Term Care Facilities—without the need to see a doctor or go to a central lab. In short: precision medicine closer to home.
Bridging the gap between discovery and practical application is a persistent challenge in research, especially when addressing complex health issues like pressure injuries. A new CIHR-funded study brings together persons affected by PI, caregivers, researchers, healthcare providers, and other stakeholders in a collaborative network. The goal is to build consensus around lived experiences, identify key challenges and prioritize strategies that can meaningfully improve prevention, management and overall health outcomes related to pressure injuries.
The team conducted 17 interviews and focus groups with people living with pressure injuries, this also included individuals with a spinal cord injury (SCI). Dr. Sharon Gabison, Principal Investigator of this study says, "there were challenges across the persona, interpersonal, organizational, and policy levels, including limited equipment access and inconsistent staffing, but also strong family support and proactive prevention practices. By surveying people with lived experience of pressure injuries and other health problems, we will be able to determine what the most urgent research and intervention priorities are."
Why It Matters
Pressure injuries can be life-threatening even for people with SCI. One example is Sepsis from pressure wounds, which continues to be a leading cause of death in Canada, despite hospital-acquired pressure injuries being labelled as "never events" in Canada.
“The conflict is clear: we know pressure injuries are deadly, yet as a system we haven’t ‘moved the needle’ in reducing them. This project aims to bridge that gap by highlighting what matters most to the people who live with and manage these injuries every day,” says Spring Hawes, Regional PLEX Engagement Liaison, Praxis. As part of this project, Spring contributed lived experience insights, helped design interview questions, and supported the development of the questionnaire.
More than just data, the project has fostered new networks of collaboration across Canada, patients, caregivers, clinicians, and researchers coming together. It has sparked conversations about why good practices aren’t universally adopted, where system breakdowns occur, and how communities can learn from what works well in certain regions.
As the next steps, the data from the survey will be reviewed by the group early next year defining consensus regarding pressure injury prevention and management. As a result, the team will shape the next phase of research, interventions, and policy advocacy.
click here to participate in the study
Praxis is pleased to support this project by bringing in the perspective of people living with spinal cord injuries, helping with knowledge translation, and committing to sharing findings more broadly.


In conversation with Praxis SCI Accelerate Cohort Member, Karma Medical (FEPSim)
About Karma Medical, one of the start-ups in the SCI Accelerate Program 2024 cohort: From a manufacturing business to MedTech innovator – How Karma Medical is changing rehabilitation technology with FEPSim
Based out of Edmonton in Canada, pair of shorts, Melissa and Darryl Short started with a general manufacturing business. The desire to diversify led them to the MedTech industry by initially developing rehab tools for stroke survivors at the Glenrose Hospital.
Things took a personal turn when the co-founders saw close family members sustaining life-altering injuries. Melissa’s brother, who lives with cerebral palsy, lost his fingers in an accident and started using Karma Medical’s early prototypes. Darryl’s brother sustained a spinal cord injury.
“We wanted to holistically try and help patients with a novel product and innovation. But it turns out we’re actually helping our family and more patients. We get to help more people. And by having family members engaged in the technology, it really helps with the success of transferring that to other patients.”
With this realization, they formed their start-up, Karma Medical Products and their flagship product, FEPSim, a hand and upper extremity rehab tool to support strength, function and independence for a dozen conditions.
Joining the Praxis SCI Accelerate Program in 2023
By 2023, FEPSim had gained traction, but to scale commercially, they were looking for guidance on business strategy and end-user feedback. That’s when they decided to join the Praxis SCI Accelerate Program with clear goals:
- Identify their target market – patients, therapists, clinics or distributors
- Commercial validation of the digital version of FEPSim
- Explore regulatory and reimbursement pathways.
What they got out of the Praxis Program
The co-leads at Karma Medical Products found access to KOLs and the insights from the Praxis external focus groups and the weekly mentorship session with the Research and PLEX (person with lived experience of SCI) advisors extremely useful:
- They learnt how to effectively engage end-users to collect meaningful feedback
- Understood how different SCI classifications can affect product usability
- Investigated how to improve long-term usability and how people with SCI would use rehab tools at home with specialized add-ons.
- Accessed resources on to understand the limits of their device’s potential reimbursement
- Conducted in-person user-testing of the digital prototype of FEPSim.
“The focus group helped us dig down a little deeper and understand how our product can serve people with SCI. It made us think differently about how we should focus on people with SCI and the different attachments or movements that they could use with the FEPSim itself.”
With momentum on their side, Karma Medical is now exploring funding opportunities, expanding their team and preparing to scale to clinics across North America. They are rooted in lived experience and driven by a mission to improve independence and recovery for people with SCI and other mobility challenges. Karma’s commitment to improving the lives of people with SCI has continued with its participation in Praxis’ SCI Adopt that funded pilots that successfully validated the functional improvements for 4 people with SCI and cost-saving for its use at the Brenda Stafford Centre & Glenrose Rehabilitation Hospital.
About the Praxis SCI Accelerate Program:
The Praxis Spinal Cord Institute’s Accelerate Program is a six-month Program geared towards prototype-stage projects with innovation to transform the lives of people with spinal cord injury (SCI). The Program provides targeted end-user product validation, research, mentorship support, tailored mentorship, expert guidance, and access to their commercialization network.


In conversation with Praxis SCI Incubate Cohort Member, smartARM
About smartARM Robotics, one of the start-ups in the SCI Incubate Program 2024 cohort: smartARM, a Toronto-based start-up co-founded by Evan Neff and Hamayal Choudhary, is an innovative start-up developing an AI-powered prosthetic arm. Their flagship device features a camera embedded in the palm to allow the arm to interpret visual cues and adapt grip functionality – enhancing the user’s ability to interact with the physical world. Evan Neff from smartARM participated in a post-program interview by the Praxis team, reflecting on the start-up’s journey, learning experiences and key takeaways from the Praxis SCI Incubate Program.
A vision for smarter prosthetics:
Evan’s interest in the human body sparked as he grew up playing sports and experiencing injuries. His interest deepened further when he saw a family member getting diagnosed with a rare form of degenerative arthritis. This led Evan to pursue MedTech engineering to help individuals recover their bodily functions.
In 2020, he met his co-founder, Hamayal Choudhary, who had been developing the concept for smartARM for a couple of years before meeting him. Their start-up, smartARM, was born out of a simple yet bold question: What if prosthetics could learn, adapt and give people true independence seamlessly?
Why smartARM joined the Praxis Incubate Program:
In April 2024, smartARM joined the Praxis Incubate Program. From the program onset, the team was clear on their goals:
- Market validation: Market exploration and end-user feedback for feasibility and alignment of their device with the needs of the SCI community.
- Clinical expertise: Ensure clinical standards and functional relevance.
- Networking and resources: Access to industry experts, mentors and commercialization experts to scale their solution.
Through the Praxis SCI Incubate Program, smartARM benefitted from the tailored support:
- User-centric design guidance from clinicians and researchers.
- End-user feedback from the SCI community through weekly mentorship sessions and focus groups.
- Connections with healthcare and commercialization experts.
With this guidance, smartARM began designing for wearability (easy to don and doff), daily usability, and therapeutic benefits (aid in rehabilitation).
Building for the future:
Initially focused on upper-arm prosthetic devices, smartARM’s mission has expanded to include exoskeletal devices and rehabilitation tools, offering hope to people with spinal cord injuries and beyond. With their vision to make assistive devices functional, accessible and user-friendly, the team aims to improve both the quality of life and the therapeutic outcomes for their users. Their journey reflects a commitment to innovation and the inclusion of the end-user perspectives in every stage of their product development and reach the markets faster.ly on. One that helps build a truly robust roadmap.”
About the Praxis SCI Incubate Program:
The Praxis Spinal Cord Institute’s Incubate Program is a four-month Program geared towards prototype stage projects with innovation to transform the lives of people with spinal cord injury (SCI). The Program provides targeted end-user product validation, research, mentorship support, tailored mentorship, expert guidance, and access to their commercialization network.


When the team at IRegained first created the MyHand® System, their goal was clear: help people regain lost hand function after a stroke or traumatic brain injury. The digital therapy platform uses targeted, personalized exercises and interactive games to support hand recovery through neuroplasticity, which is the brain’s inherent ability to reprogram its neural pathways and relearn.
The innovation showed early promise, and its potential expanded significantly after a meeting with Praxis, where the IRegained team was introduced to how the MyHand® System could benefit the spinal cord injury (SCI) community. Recognizing this untapped opportunity, Praxis supported IRegained through its inaugural SCI Adopt program following a thorough evaluation process and began working closely with the team to adapt the system for people living with SCI. More than just a funding partner, Praxis offered access to a community of clinicians, people with lived experience, and neurorehabilitation specialists who helped shape the product’s evolution in real-time.
“We originally focused on stroke because that’s where the main target population, funding and research were,” says Samir Sahoo, Chief Operating Officer, IRegained. “Praxis expanded our vision. They helped us see a real, unmet need in the SCI community, and they gave us the tools and connections to do something about it.”
One of the most important early insights came from user experience. The team had built the system for one-hand impairment, as this is common for people who have had a stroke. After meeting SCI users with bilateral hand impairments, the IRegained team quickly learned that their technology needed to adapt to have a greater impact. “It seems so simple now,” adds Eric Dumais, Deputy Director, Clinical Operations, IRegained. “Designing for both hands changed everything. The feedback from Praxis made the system more accessible, inclusive, and ultimately more effective.”
As part of the program, Praxis funded the deployment of MyHand® at Walk-It-Off Neuro-Recovery and Wellness Centre, a rehabilitation clinic in Newmarket, Ontario. Over a three-month pilot, five individuals living with SCI used the MyHand® System, and the results were both encouraging and moving. Patients reported increased grip strength and functional improvements. Even more powerfully, they described regaining a sense of independence in their daily lives.
“We are very thankful to Praxis for generously funding a MyHand® System for Walk It Off. Clinicians enjoy it and find it intuitive to use with patients, while patients who use it find it fun and engaging.”
– K., RKin at Walk-It-Off.
“We previously met someone with a C4-C5 injury who had very limited hand function,” Dumais adds. “Seeing even small gains in their ability to interact with the world, it was emotional. That’s when we knew we were on the right path.”
Working with Praxis’ Dr. Vanessa Noonan and the persons with lived experience team, IRegained identified a cost-effective and meaningful way to measure outcomes. Grip strength, user feedback, and simple questionnaires replaced more expensive and time-consuming clinical assessments, ensuring valuable insights were compiled without overwhelming clinics.
Praxis also played a key role in supporting a health economics analysis, providing data on quality-adjusted life years, standards of care, and policy trends. With this foundation, IRegained built a compelling case: their device could offer equal or better clinical outcomes than current approaches, at a lower long-term cost. Early data suggests potential savings of around $160 per patient per year, along with reduced burden on clinicians, and even potentially reduced reliance on caregivers.
“There’s a clear benefit to using our technology,” Sahoo says. “The question is, if not MyHand®, then what? Clinics are often left with tools that don’t make a meaningful difference. MyHand® offers the potential for real gains in function and overall quality of life.”
Looking ahead, IRegained’s goal is to expand access and adoption, it is currently available at three locations in Canada and four in the US. “Everyone who uses the device wants to keep using it. It’s our mission to bring this technology to more people, more clinics, and more communities,” says Sahoo.
Both Sahoo and Dumais credit Praxis for the critical role it has played in IRegained’s journey. “No other organization does what Praxis does, connecting companies like ours with the networks and people we want our product to impact. Without the support of Praxis, MyHand® wouldn’t be where it is today. And neither would we,” says Dumais.

IRegained received support from Praxis as part of the inaugural SCI Adopt program, an initiative designed to advance innovative solutions in spinal cord injury care.